Solutions
Our easy-to-use, secure SaaS solutions help sites, site networks, integrated research organizations, CROs and sponsors stay connected, optimized and aligned.
For Clinical Research Sites
For Clinical Operations Teams
Overview
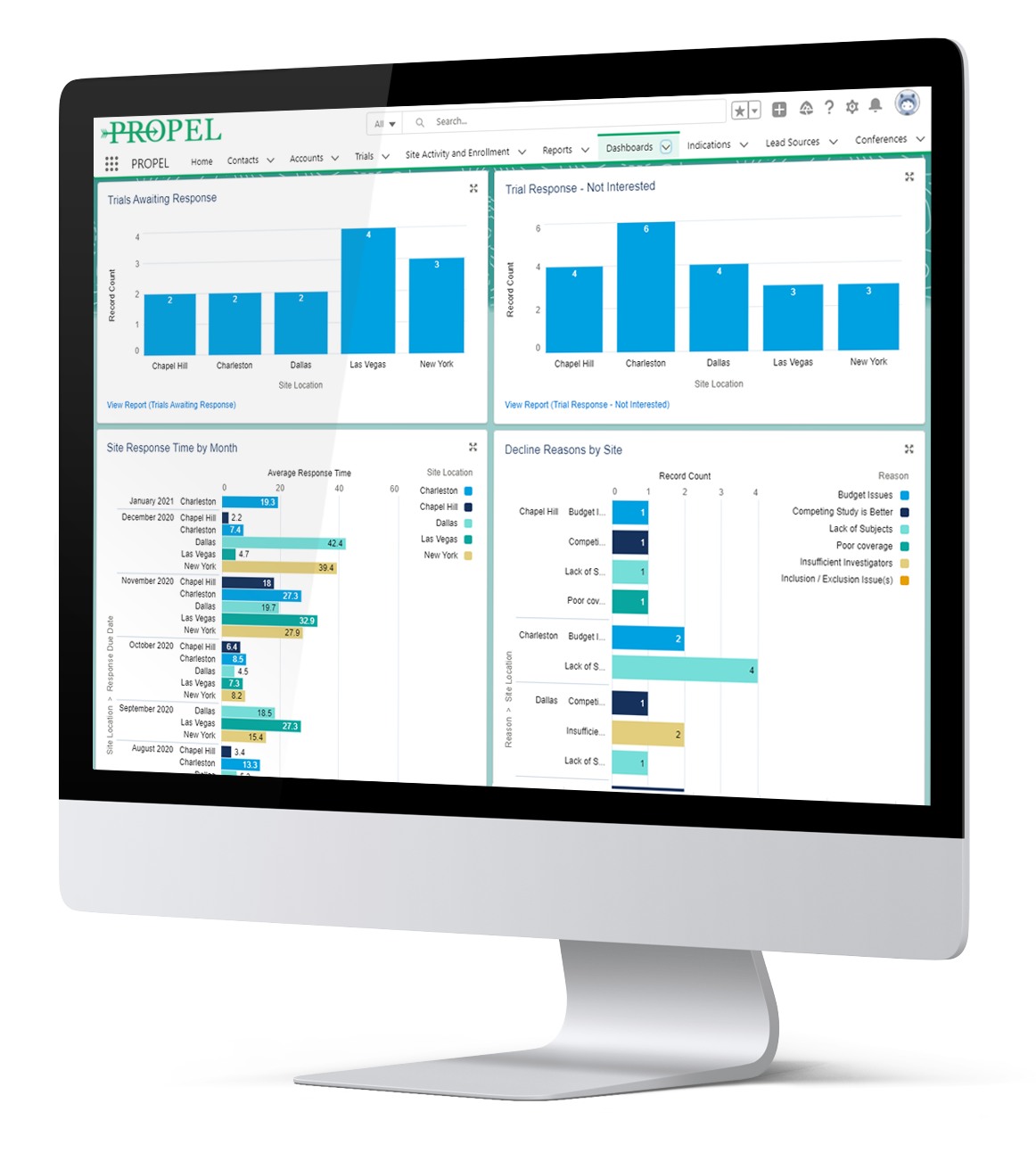
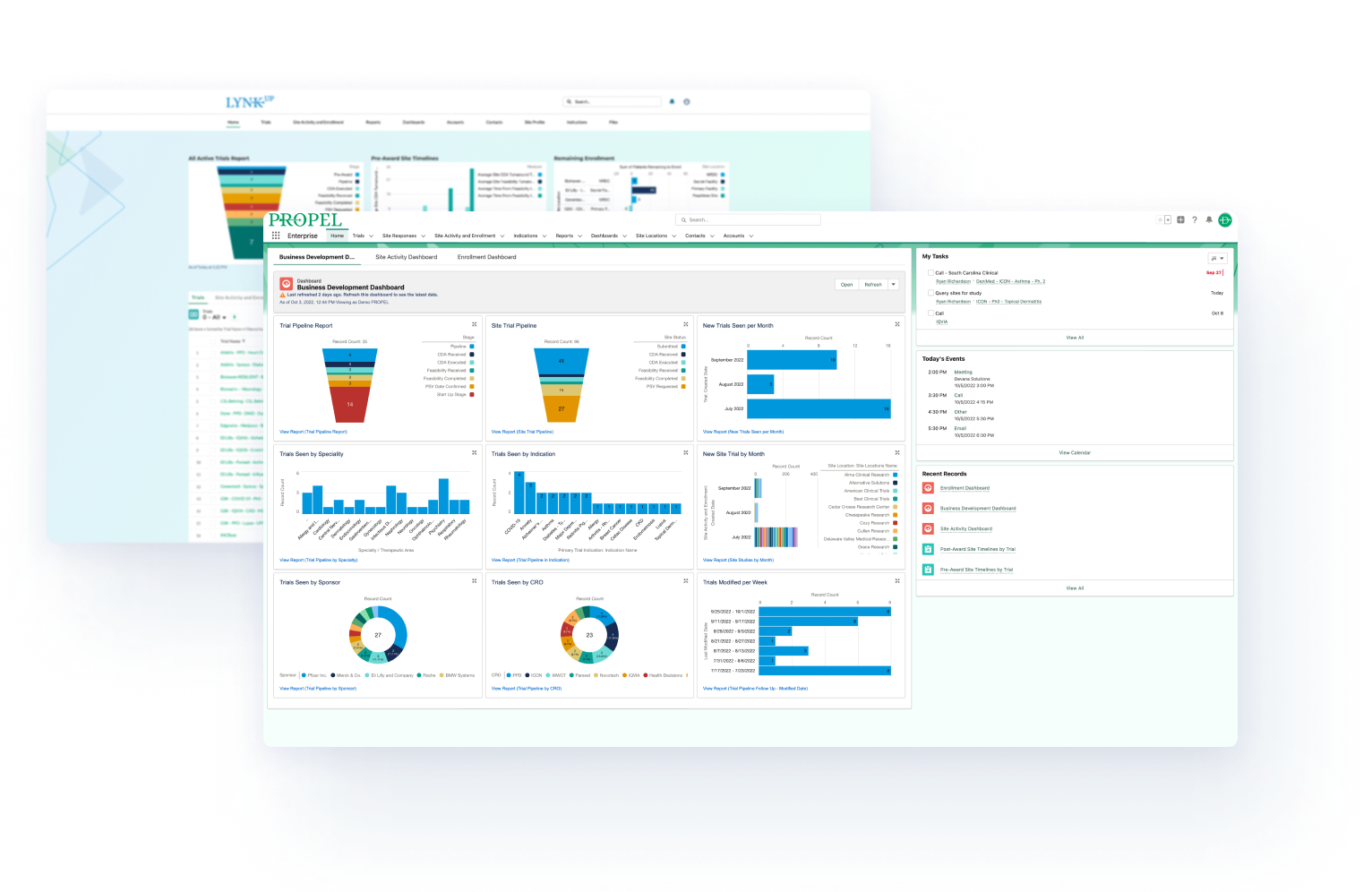
Share and analyze performance data in real time and across all stakeholders—from site-level to CRO to sponsor—to track trial progress, make better decisions and increase your chances of a successful outcome.
Automate pipeline management, study startup workflows, and securely connect all functional groups in your organization.
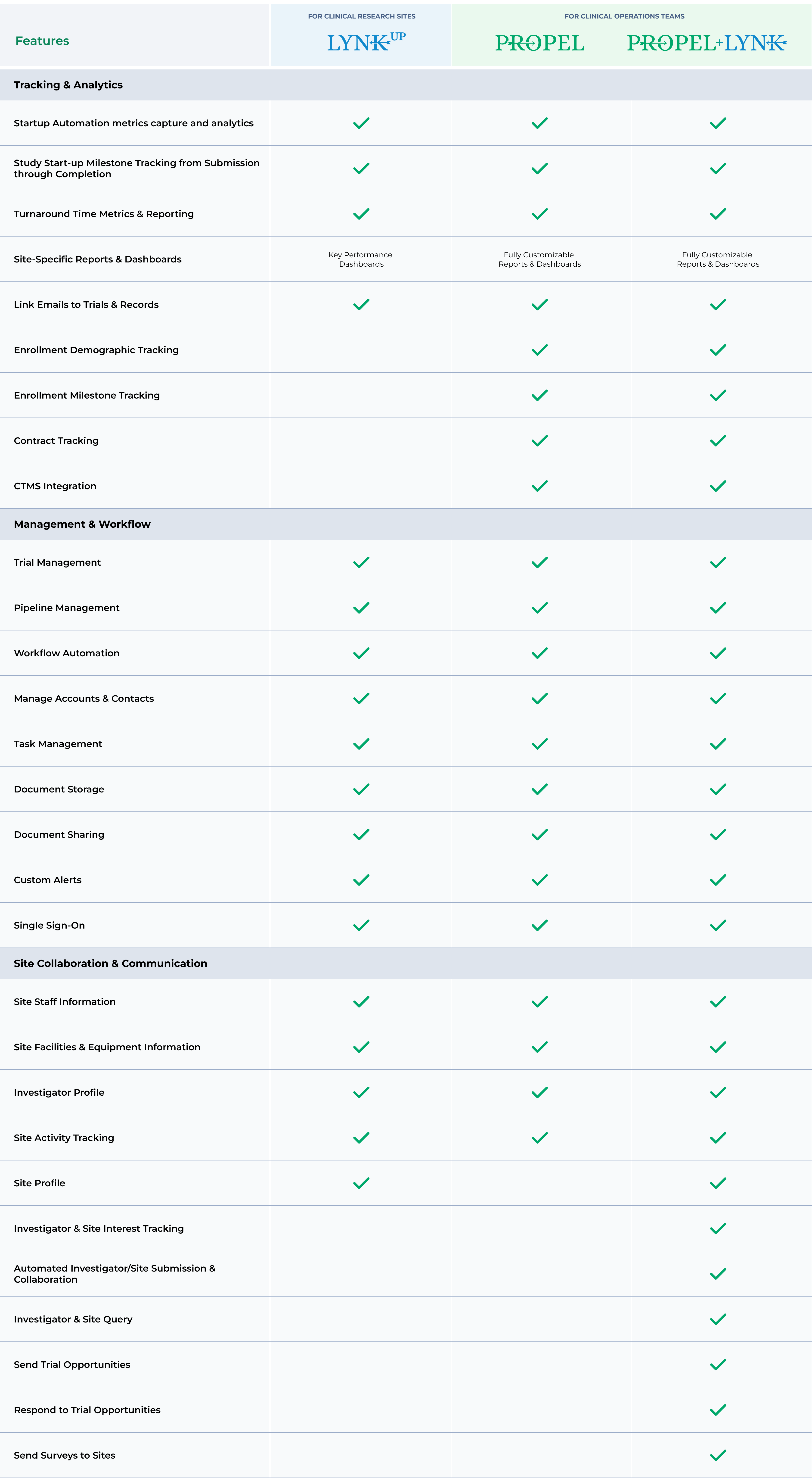
Integrate your CTMS with PROPEL so that key data points entered into either system are automatically synchronized across both platforms. Learn More >
Automate start-up, analytics & reporting across your research organization.
-
Study Startup workflow automation and end-to-end trial performance data capture, aggregation, and analysis for solid data-driven decision-making
-
Ability to link multiple contacts, documents, automate reminders, assign tasks and capture communication across every clinical trial for time savings and performance transparency
-
Performance data transparency across all functional teams and stakeholders to drive accountability
-
Cloud-based for remote access yet highly secure to connect and align decentralized study teams